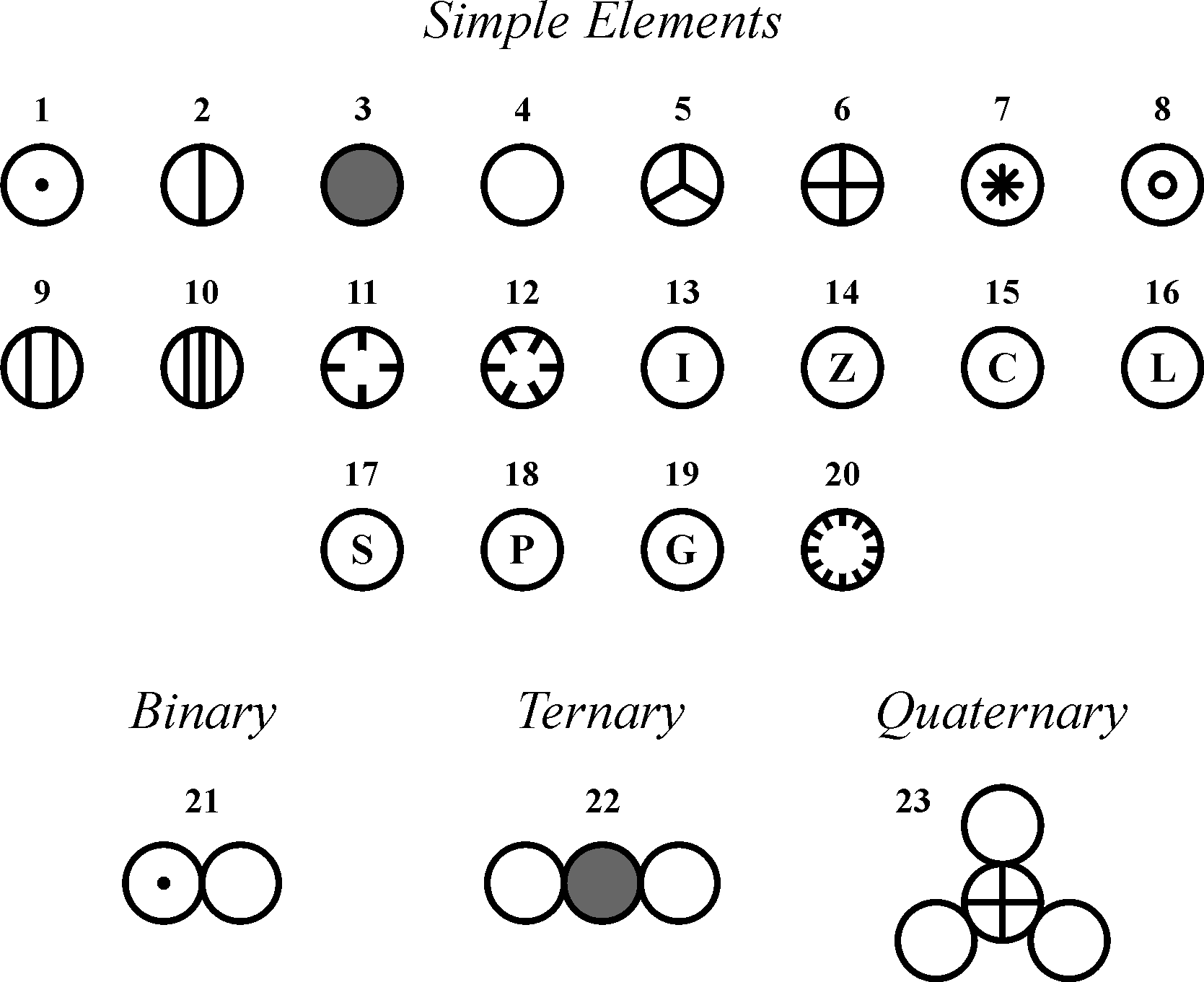
If a substance is burnt, phlogiston evolves and goes into atmosphere and clax (meaning a non-useful thing) is left behind as an ash. Daltons atomic theory also stated that all compounds were composed of combinations of these atoms in defined ratios.
While all atoms of an element were identical, different elements had atoms of differing size and mass.

The theory stated that the substance undergoing combustion mainly contains a mysterious matter called phlogiston and some clax. Daltons atomic theory proposed that all matter was composed of atoms, indivisible and indestructible building blocks. Composition: Democritus believed that atoms were indivisible, while Dalton proposed that atoms had a fixed atomic weight and could be broken down into smaller particles. changeĭuring chemical change both physical and chemical properties of the substance change.Įxamples: Freezing of water, melting of ice, Heating of tungsten wire to red hot condition.Įxamples: Rusting of iron, burning of wood, digestion of food, etc. The atomic theory explains the laws of chemical combination (the Law of Constant Composition and the Law of Multiple Proportions ). Origin: Democritus’ atomic theory was based on philosophical reasoning, while Dalton’s atomic theory was based on empirical evidence. There is enthalpy change in chemical change.ĭuring physical change physical properties like phase, texture, shape, size, etc. There is negligible change in enthalpy of the system in physical change. In a chemical change, energy is produced (heat, light, sound, etc.) For example, if an element such as copper consists of only one kind of atom, then it cannot be broken down into simpler substances, that is, into substances composed of fewer types of atoms.

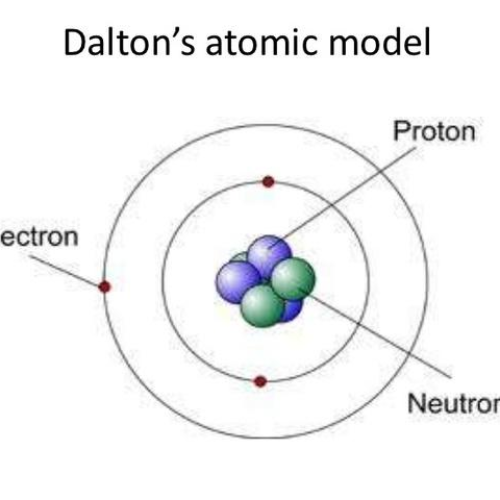
In a physical change, no energy is produced. Dalton’s atomic theory provides a microscopic explanation of the many macroscopic properties of matter that you’ve learned about. Physical change is reversible and original substance can be recovered.Ĭhemical change is irreversible and original substance cannot be recovered. The matter is made up of indivisible particles known as atoms. The molecular composition of a substance changes completely Many consider 2008 the 200th anniversary of atomic theory, John Dalton’s momentous theory of the nature of matter. We know now that (1) an atom can be further subdivided, (2) all atoms of an element are not identical in mass, and (3) using nuclear fission and fusion techniques, we can create or destroy atoms by changing them into other atoms. There is only rearrangement of molecules while their actual composition remains same. Daltons atomic theory has been largely accepted by the scientific community, with the exception of three changes. Daltons Atomic Theory is the first scientific theory to relate chemical changes to the structure, properties, and behavior of the atom. Physical changes are those changes in which there is an only change in the appearance of the substance but no change in its chemical composition.Ĭhemical changes are those changes which cause a change in chemical composition of a substance.Įntirely new substance (one or more) is formed. Trending Questions How do gases fill a container? How does the energy source coal work? How much does 1 Cubic meter of Compressed Natural Gas weight in kilograms? How many carbon atoms are present in a mole of 12c using eight significant figures? Is 4f a valid orbital shell designation? What things are sold in groups of 11 or come in groups of 11? What is the Balanced equation for magnesium sulfate ana lead nitrate? What is it called when water vapor turns from a gas to a liquid? Is glass absorbent? A steel cylinder has a length of 2.16 a radius of 0.Distinguishing Between Physical Changes and Chemical Changes: Physical Change

